Transcranial Magnetic Stimulation (TMS)
TMS Therapy is an FDA approved revolutionary breakthrough in the treatment of depression
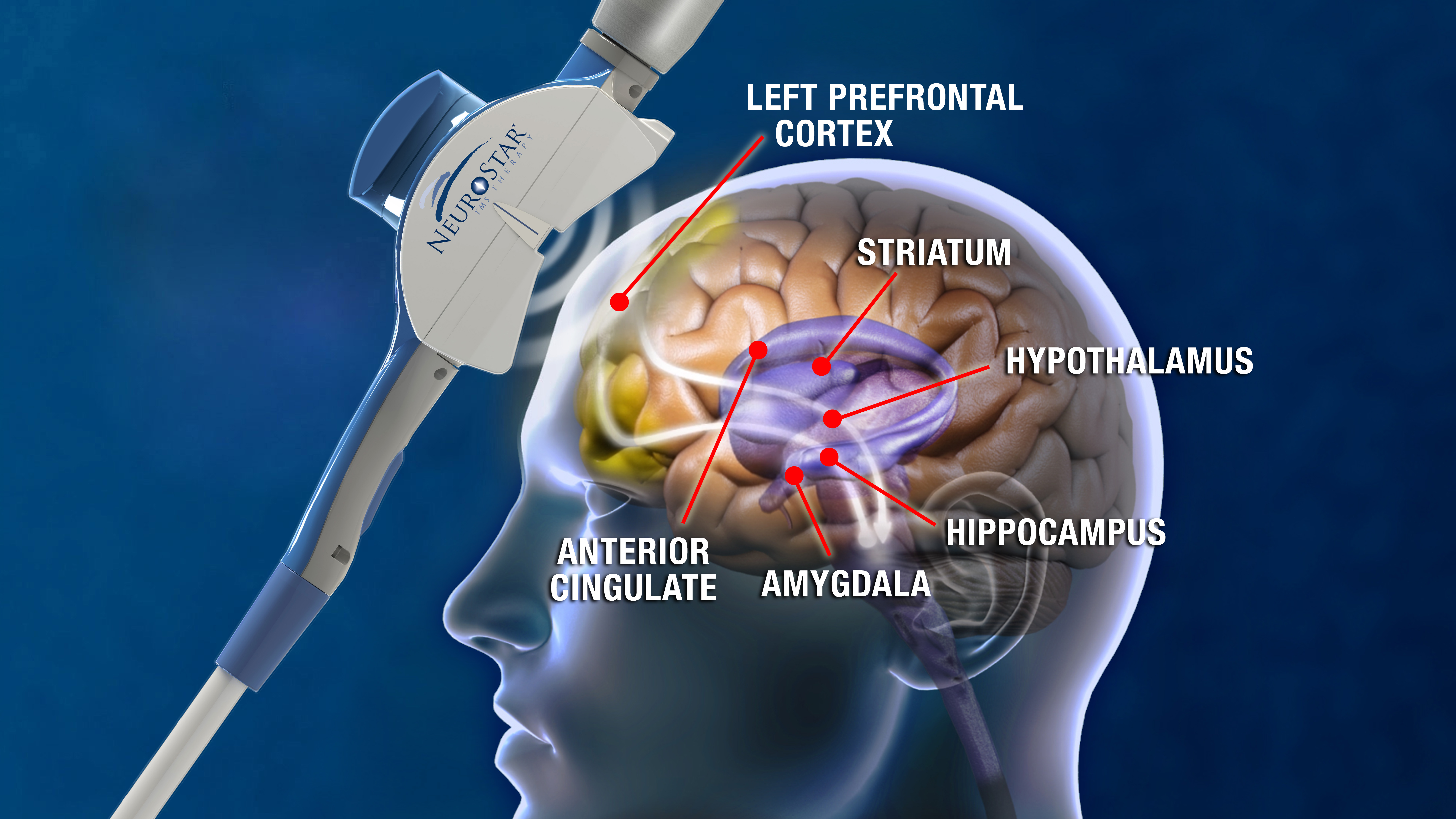
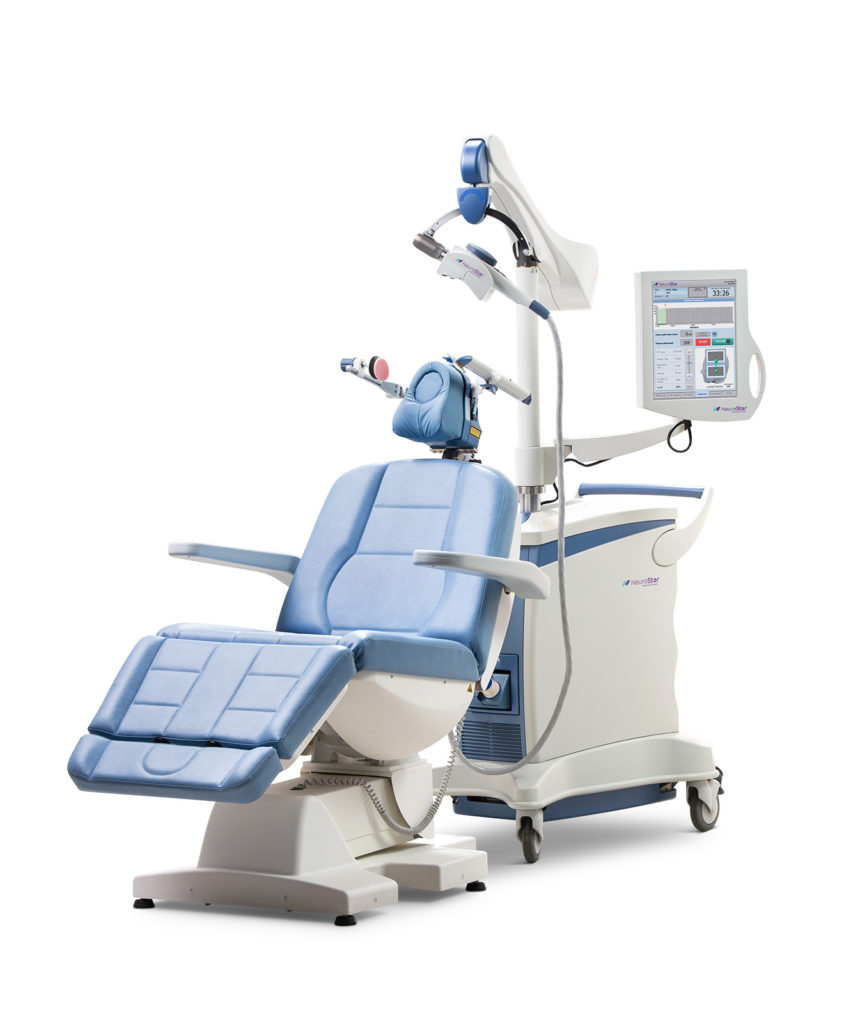
Transcranial Magnetic Stimulation (rTMS) is a technological breakthrough in the treatment of Major Depression. Cleared for use by the FDA in October 2008, TMS is a non-invasive, non-systemic treatment that utilizes MRI strength magnetic pulses to stimulate areas of the brain known to be hypoactive in patients with depression. TMS is a proven and highly successful treatment for Major Depressive Disorder in adult patients who have failed to receive satisfactory improvement from prior antidepressant medication.
TMS is an effective alternative treatment for Major Depressive Disorder and because TMS is non-systemic (not absorbed throughout the body as medications are), patients may resume their normal daily activity after treatments.
No sedation is necessary prior to TMS treatment and side effects are minimal. Some patients may experience slight pain or a headache near the area of treatment. If patients experience discomfort, they can take an over the counter analgesic such as ibuprofen or Tylenol.
Since no anesthesia is needed, patients are awake and alert during the treatment. They can easily resume normal activities afterward. Each TMS treatment session lasts between 20 to 30 minutes and are typically given 5 days a week for 6 weeks, followed by a 2-3 week taper period.
Many commercial health plans as well as Medicaid and Medicare cover TMS treatments. If you are a candidate for TMS, our office will contact your insurance company and request prior authorization on your behalf. All potential TMS patients must undergo an in-office psychiatric assessment prior to initiation of treatment.
To schedule an appointment for a TMS assessment, please call our office at 614-580-6917.

Facts about Depression
Many creative individuals experienced depression, including Ludwig van Beethoven, John Lennon, Edgar Allan Poe, Mark Twain, Georgia O’Keefe, Vincent van Gogh, Ernest Hemmingway, F. Scott Fitzgerald and Sylvia Plath.