ABOUT TMS THERAPY
What actually is transcranial magnetic stimulation or TMS therapy?
How can it help with my depression?
Transcranial Magnetic Stimulation or TMS:
An FDA Approved Treatment Option For Depression.
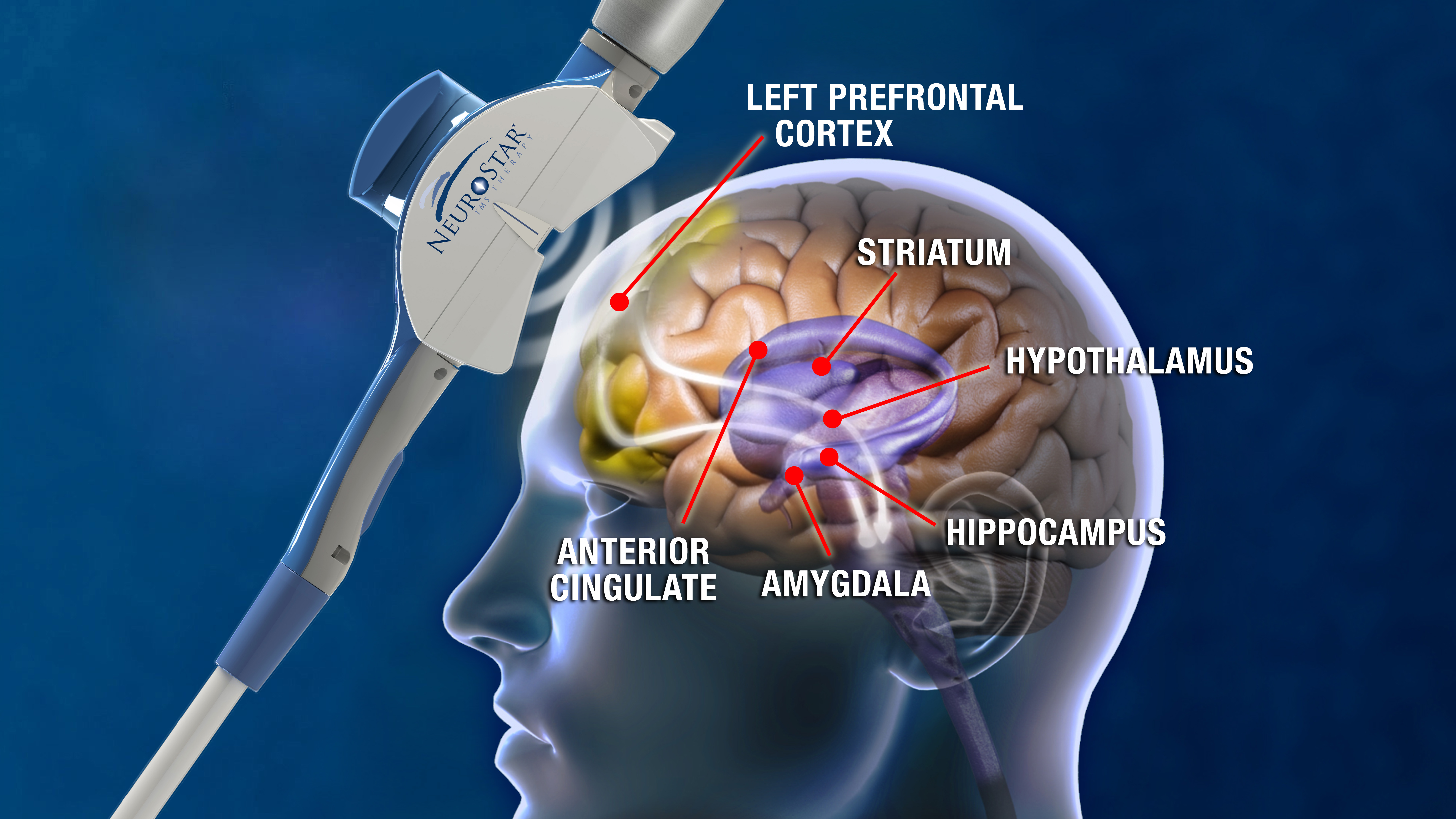
Transcranial Magnetic Stimulation (rTMS) is a technological breakthrough in the treatment of Major Depression. Cleared for use by the FDA in October 2008, TMS is a non-invasive, non-systemic treatment that utilizes MRI strength magnetic pulses to stimulate areas of the brain known to be hypoactive in patients with depression. TMS is a proven and highly successful treatment for Major Depressive Disorder in adult patients who have failed to receive satisfactory improvement from prior antidepressant medication.
TMS is an effective alternative treatment for Major Depressive Disorder and because TMS is non-systemic (not absorbed throughout the body as medications are), patients may resume their normal daily activity after treatments.
No sedation is necessary prior to TMS treatment and side effects are minimal. Some patients may experience slight pain or a headache near the area of treatment. If patients experience discomfort, they can take an over the counter analgesic such as ibuprofen or Tylenol.
Since no anesthesia is needed, patients are awake and alert during the treatment. They can easily resume normal activities afterward. Each TMS treatment session lasts between 20 to 30 minutes and are typically given 5 days a week for 6 weeks, followed by a 2-3 week taper period.
Many commercial health plans as well as Medicare cover TMS treatments. If you are a candidate for TMS, our office will contact your insurance company and request prior authorization on your behalf. All potential TMS patients must undergo an in-office psychiatric assessment prior to initiation of treatment.
To schedule an appointment for a TMS assessment, please call our office at 614-580-6917.
 Is TMS therapy right for me?
Is TMS therapy right for me?
While the American Psychiatric Association has stated that they would consider using TMS therapy after one failed trial of a first line antidepressant medication, most insurance companies require 3-4 failed trials of antidepressant medications prior to authorizing TMS therapy. Most patients that we have worked with feel like, “I’ve tried everything and nothing is working.”
If there is a question as to whether TMS would be appropriate for you, we recommend scheduling an evaluation for TMS therapy to consider the options available to you.
Have you tried more than three antidepressants without remission of your depressive symptoms?
Have you been unable to tolerate your antidepressant medications?
Currently, are you on more than one anti-depressant medication?
Is your current medication failing to provide adequate benefit for your depression?
Do the side effects of your medication outweigh the benefit?
Has depression increased your doctor visits for other healthcare conditions?
Is depression affecting your ability to function in your life including work, school, taking care of yourself or your family obligations?
Clinic studies show that the response rate to TMS therapy exceeds that of medication-based treatments. These studies were of patients who had not responded to multiple trials of antidepressant medications and psychotherapy.
The evidence for the clinical efficacy of TMS in the treatment of depression is considerable, spanning more than 30 controlled clinical research studies, including NIMH sponsored studies.
Easily tolerated. The most common side effect is mild scalp pain or discomfort.
No anesthesia or adverse effects on memory. No change in daily activities.
Adopted by insurers covering 216 million US lives including UBH, Aetna, Medical Mutual, Anthem, Medicare, and many others. Medicaid does not cover TMS therapy at this time. It is also possible to self-pay.







A TMS Experience
I am currently 44 years old and have suffered from depression since elementary school. I spent the majority of my older kids’ early childhood in bed, in the dark. Through past attempts to feel joy and experience life, I have been on several different meds. Some didn’t work at all. Some had very unpleasant side effects. Some made me feel better initially, then the dose had to be increased. Repeat.
I decided to give TMS treatment a try because what did I have to lose? I was in a dark, dark place during my initial visit with Dr. Yoho. All I wanted to do was sleep. However, my three children and associated parental obligations prevented me from indulging my depression in this manner. Every day was a struggle. My depression manifested in fatigue, anger, irritability, moodiness, you name it. Ultimately, I decided to visit Emerald Psychiatry because my depression was becoming highly detrimental to my family.
Initially, I was concerned about side effects. Could TMS cause permanent brain damage? Could TMS change my personality? Could TMS render me a vegetable? Is the risk worth the reward? Prior to beginning treatment, I researched TMS via NIMH (National Institute of Mental Health) and PubMed. The research, which indicated little to no side effects and a high percentage of positive outcomes, validated my decision to move forward.
TMS therapy did not bother me at all. I would just close my eyes and picture each little pulse to be a beam of light/happiness absorbing into my brain. Somewhere around the 4th week of therapy, I began to notice that I was feeling better. I had no desire to lay in bed; I was not quick to anger. I enjoyed interacting with my kids and husband, and I became much more patient with people and situations in general. Any thoughts of not wanting to exist had completely disappeared. It seemed like I had become “excited about life” for the first time in memory.
I would absolutely recommend TMS to relatives or friends experiencing depression to the degree that I was. I feel so much better. I have my life back, I can experience joy. I am a better mom and wife and friend (I’m in the process of organizing a girls’ trip, which I never would have done).
-Stephanie B.